
Article | April 25, 2024
Using Personal Data to Train AI? Make Sure You Comply with State Requirements
Read More

Article | April 23, 2024
Senators Urge FDA: Fix Ad Oversight
Read More

Article | April 16, 2024
Amidst Legal Fight, HHS Revises Online Tracking Guidance
Read More
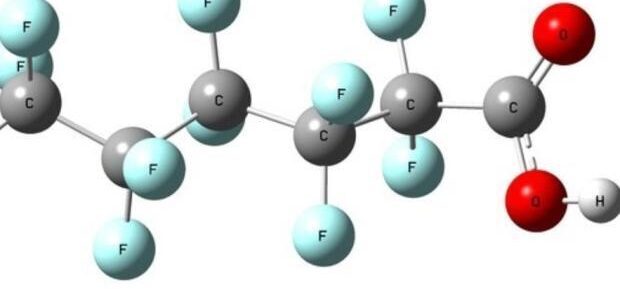
Article | April 09, 2024
What Manufacturers Need to Know About PFAS
Read More

Article | April 04, 2024
FDA Issues Supplemental Draft Guidance for Premarket Cybersecurity
Read More

Article | April 02, 2024
FDA's View on Cannabis
Read More

Article | March 27, 2024
Record Year for the DOJ: FCA Enforcement Trends
Read More

Article | March 21, 2024
FDA Finalizes New QMSR for Medical Device Manufacturers
Read More

Article | March 18, 2024
FDA Untitled Letter Targets Misleading Drug Advertising
Read More

Article | March 14, 2024
Nevada Filing Deadlines are Approaching: What Manufacturers Need to Know
Read More

Article | March 12, 2024
FDA Issues Numerous Warning Letters
Read More

Article | March 06, 2024
CCPA Regulations to Take Effect Immediately
Read More












